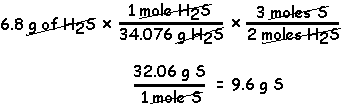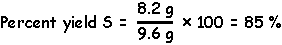In a balanced equation, the coefficients represent the number of atoms of each element in the products and reactants. The coefficients also represent the number of molecules and moles in the products and reactants.
It is sometimes mistakenly believed that reactions follow out to completion using up all of the reactants or limiting reactants. However, chemical reactions do not always act so conveniently, and reactions, called side reactions, can use up some of the reactants.
The actual amount of product produced divided by the theoretically maximum amount of product which could be produced (100%) is called the percent yield.
Example #1:
The reaction of 6.8 g of H2S with excess SO2 according to the following reaction yields 8.2 g of S. What is the percent yield?
(Atomic weights: H = 1.008, S = 32.06, O = 16.00).
2 H2S + SO2 3 S + 2 H2O
In the above reaction, 2 moles H2S react to produce 3 moles S.
1) Use stoichiometry to determine the maximum amount of S that could be produced with 6.8 g of H2S.
2) Divide the actual amount by the theoretical maximum amount of S produced in this reaction and multiply by 100.
Percent Yield
In a balanced equation, the coefficients represent the number of atoms of each element in the products and reactants. The coefficients also represent the number of molecules and moles in the products and reactants.
It is sometimes mistakenly believed that reactions follow out to completion using up all of the reactants or limiting reactants. However, chemical reactions do not always act so conveniently, and reactions, called side reactions, can use up some of the reactants.
The actual amount of product produced divided by the theoretically maximum amount of product which could be produced (100%) is called the percent yied.
Example #1:
The reaction of 6.8 g of H2S with excess SO2 according to the following reaction yields 8.2 g of S. What is the percent yield?
(Atomic weights: H = 1.008, S = 32.06, O = 16.00).
2 H2S + SO2 3 S + 2 H2O
In the above reaction, 2 moles H2S react to produce 3 moles S.
1) Use stoichiometry to determine the maximum amount of S that could be produced with 6.8 g of H2S.
2) Divide the actual amount by the theoretical maximum amount of S produced in this reaction and multiply by 100.
Modern view of atomic structure
The Modern View of Atomic Structure
Physicists have identified a long list of particles which make up the atomic nucleus. Chemists, however, are primarily concerned with the following sub-atomic particles:
- electron
- proton
- neutron
The electron is negatively charged, with a charge of -1.602 x 10-19 Coulombs (C). For convenience, the charge of atomic and sub-atomic particles are usually described as a multiple of this value (also known as the electronic charge). Thus, the charge of the electron is usually simply refered to as -1.
Proton
The proton has a charge of +1 electron charge (or, +1.602 x 10-19 C)
Neutrons
Neutrons have no charge, they are electrically neutral.
Note: Because atoms have an equal number of electrons and protons, they have no net electrical charge
Protons and neutrons are located in the nucleus (center) of the atom. The nucleus is small compared to the overall size of the atom. The majority of the space of an atom is the space in which the electrons move around.
Electrons are attracted to the protons in the nucleus by the force of attraction between particles of opposite charge.
Note: The strength of attraction between electrons and protons in the nuclei for different atoms is the basis of many of the unique properties of different atoms. The electrons play a major role in chemical reactions. In atomic models, the electrons are represented as a diffuse electron cloud
The mass of an atom is extremely small. The units of mass used to describe atomic particles is the atomic mass unit (or amu).
An atomic mass unit is equal to 1.66054 x 10-24 grams.
How do the different sub-atomic particles compare as far as their mass?
Proton = 1.0073 amu
Neutron = 1.0087 amu
Electron = 5.486 x 10-4 amu
From this comparison we can see that:
- The mass of the proton and neutron are nearly identical
- The nucleus (protons plus neutrons) contains virtually all of the mass of the atom
- The electrons, while equal and opposite in charge to the protons, have only 0.05% the mass
Note: a convenient unit of measurement for atomic distances is the angstrom (Å). The angstrom is equal to 1 x 10-10 meters. Thus, most atoms are between 1 and 5 angstroms in diameter.
Pinheads and Bullion Cubes
Pinheads have a diameter of about 1 x 10-3 meters (a millimeter across). If an atom had a diameter of 2.5 x 10-10 meters, then
(1 atom/2.5 x 10-10 meters) * (1 x 10-3 meters) = 4 x 106 atoms
i.e. four million of them could line up across the head of a pin.
The diameter of atomic nuclei are about 10-4Å. Thus, the nuclei is about 0.01% the diameter of the atom as a whole. If the nucleus had a diameter equal to that of a pinhead, then the atom itself would have a diameter of some 10 meters (about 39 and a half feet).
The nucleus of an atom is therefore quite dense. Consider a simple case of a nucleus containing 1 neutron and 1 proton:
mass of nucleus = ~2.0 amu = 2 * (1.66 x 10-24 grams) = 3.32 x 10-24 grams
diameter of nucleus = (approximately) 1 x 10-4 Å = 1 x 10-14 meters
radius of nucleus = 1 x 10-14 meters/2 = 0.5 x 10-14 meters
volume of nucleus = (4/3)p(radius of nucleus)3
volume of nucleus = 5.24 x 10-43 meters3
mass/volume = 3.32 x 10-24 grams/5.24 x 10-43 meters3
mass/volume = 6.34 x 1018 grams/meter3
a bouillon cube (stuff you can make soup out of if you are really broke) is about one cubic centimeter, or 1 x 10-6 meters3 and if it were made up of atomic nuclei it would weigh:
(1 x 1018 grams/meter3) * (1 x 10-6 meters3) = 6.34 x 1012 grams
or about six billion kilograms, or about 2.8 billion tons.
Isotopes, Atomic Numbers and Mass Numbers
What characteristic feature of sub-atomic particles distinguishes one element from another?
- All atoms of an element have the same number of protons in the nucleus
- Since the net charge on an atom is 0, the atom must have an equal number of electrons.
- What about the neutrons? Although usually equal to the number of protons, the number of neutrons can vary somewhat. Atoms which differ only in the number of neutrons are called isotopes. Since the neutron is about 1.0087 amu (the proton is 1.0073), different isotopes have different masses.
All atoms of the element Carbon (C) have 6 protons and 6 electrons. The number of protons in the carbon atom are denoted by a subscript on the left of the atomic symbol:
This is called the atomic number, and since it is always 6 for carbon, it is somewhat redundant and usually omitted. Another number, the "Mass Number" is a superscript on the left of the atomic symbol. It denotes the sum of the number of protons and neutrons in the particular isotope being described. For example:
refers to an isotope of carbon which has (as expected for the element carbon) six protons, and six neutrons. The following isotope of carbon:
has 6 protons (atomic number) and 8 neutrons (8=14-6). This isotope is also known simply as "carbon 14". Carbon 12 is the most common form of carbon (~99% of all carbon). An atom of a specific isotope is called a nuclide.
Since all atoms are composed of protons, electrons and neutrons, all chemical and physical differences between elements are due to the differences in the number of these sub-atomic particles. Therefore, an atom is the smallest sample of an element, because dividing an atom further (into sub-atomic particles) destroys the element's unique identitity.
Measurement
Unit Conversions
- Converting Units with Conversion Factors
- Using Multiple Conversion Factors - Part 1
- Using Multiple Conversion Factors - Part 2
- Understanding Conversion Factors
- Unit Conversions with Area and Volume
- Converting Metric Units of Volume
Chapter Lectures (times in parentheses)