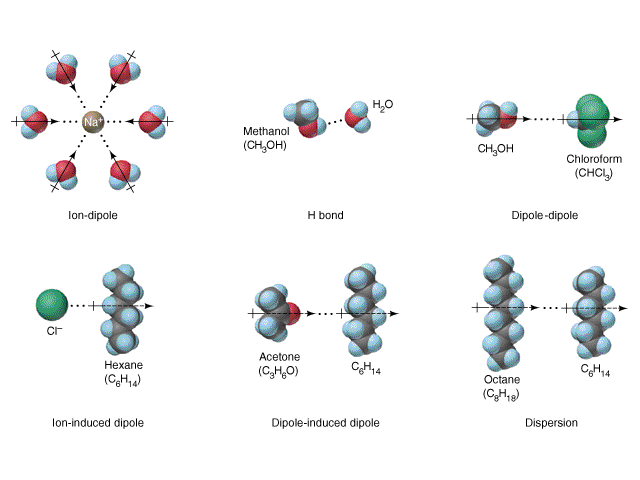
Intermolecular Forces (IMFs)
Image retrieved from http://www.science.uwaterloo.ca/~cchieh/cact/fig/interaction.gif
So, just what are IMF's?
The physical properties of condensed matter (liquids and solids) can be explained in terms of the kinetic molecular theory. In a liquid, intermolecular attractive forces hold the molecules in contact, although they still have sufficient KE to move past each other.
Intermolecular attractive forces, collectively referred to as van der Waals forces, are responsible for the behavior of liquids and solids and are electrostatic in nature. Dipole-dipole attractions result from the electrostatic attraction of the partial negative end of one dipolar molecule for the partial positive end of another. The temporary dipole that results from the motion of the electrons in an atom can induce a dipole in an adjacent atom and give rise to the London dispersion force. London forces increase with increasing molecular size. Hydrogen bonds are a special type of dipole-dipole attraction that results when hydrogen is bonded to one of the three most electronegative elements: F, O, or N.
(Text from openstax Chemistry - Download for free at http://cnx.org/content/col11760/latest/)
GenChem Level Videos
Liquids and Intermolecular Forces: Part 1 of 10 [8:39] by Dr. Mike Christiansen
In this video Dr. Christiansen reviews the differences between solids, liquids, and gases.He'll also teach you about dipole-dipole forces and hydrogen bonding.
Liquids and Intermolecular Forces: Part 2 of 10 [8:58] by Dr. Mike Christiansen
In this video Dr. Christiansen will teach you about ion-dipole and dispersion forces (also called London or Van der Waals) forces.
Intermolecular Forces [6:58] by Fus Chemistry Videos
Types of Intermolecular Forces [8:06] by Fus Chemistry Videos
Solids and Liquids [7:28] by Bozeman Science
In this video Paul Andersen compares and contrasts the properties of solids and liquids. Solids have a more organized structure which can either be amorphous or crystalline. In liquids the intermolecular forces are lower and so the molecules can show translation. Some of the properties that can be observed in liquids are viscosity, surface pressure and volumes of mixing.
London Dispersion Forces [5:01] by Bozeman Science
In this video Paul Andersen describes the positive force intermolecular forces found between all atoms and molecules. As electrons are distributed unevenly it creates instantaneous dipoles which hold molecules together. This force even holds uncharged atoms (like Noble gases) together. London dispersion forces increase as surface area, molecule sizing and pi bonding increases.
Dipole Forces [5:01] by Bozeman Science
In this video Paul Andersen describes the intermolecular forces associated with dipoles. A dipole is a molecule that has split charge. Dipole may form associations with other dipoles, induced dipoles or ions. An important type of dipole-dipole forces are hydrogen bonds.
Intermolecular Forces [7:00] by Bozeman Science
In this video Paul Andersen explains how intermolecular forces differ from intramolecular forces. He then explains how differences in these forces account for different properties in solid, liquids and gases. Some of these properties include the boiling point, melting point, surface tension, capillary action and miscibility. Intermolecular forces between gas molecules creates variation from ideal gas law.
Introductory Level Videos
Polar & Non-Polar Molecules: Crash Course Chemistry #23 [10:45] by CrashCourse
Molecules come in infinite varieties, so in order to help the complicated chemical world make a little more sense, we classify and categorize them. One of the most important of those classifications is whether a molecule is polar or non-polar, which describes a kind of symmetry - not just of the molecule, but of the charge. In this edition of Crash Course Chemistry, Hank comes out for Team Polar, and describes why these molecules are so interesting to him.
You'll learn that molecules need to have both charge asymmetry and geometric asymmetry to be polar, and that charge asymmetry is caused by a difference in electronegativities. You'll also learn how to notate a dipole moment (or charge separation) of a molecule, the physical mechanism behind like dissolves like, and why water is so dang good at fostering life on Earth.
Liquids: Crash Course Chemistry #26 [11:03] by CrashCourse
In this episode of Crash Course Chemistry, Hank gives you the low down on things like London Dispersion Forces, Hydrogen Bonds, Cohesion, Adhesion, Viscosity, Capillary Action, Surface Tension, and why liquids are just... WEIRD!
Intermolecular Forces and Boiling Points [10:53] by Professor Dave Explains
Why do different liquids boil at different temperatures? It has to do with how strongly the molecules interact with each other. Find out all the different ways, and how to use them to make predictions about matter!
Intermolecular Forces [9:15] by The Chemistry Solution
This chemistry tutorial is designed to help compare and contrast commonly encountered intermolecular forces and interactions. The intermolecular forces that are discussed are ion-dipole interactions, dipole-dipole interactions, hydrogen bonding interactions and London dispersion forces.
Additional Useful Resources
Intermolecular Properties - web-based tutorials for GenChem at The University of Texas
Forces of Attraction - web-based tutorials for GenChem at The University of Texas
Practice
Forces of Attraction - link to webpage with link to worksheet and answer key for GenChem at The University of Texas
The Solid and Liquid States - Quiz bank for GenChem at Ohio State
"Click" on the appropriate "Quiz" button to select quizzes for practice from the following topics:
Liquids and Intermolecular Forces: Part 1 of 10 [8:39] by Dr. Mike Christiansen
In this video Dr. Christiansen reviews the differences between solids, liquids, and gases.He'll also teach you about dipole-dipole forces and hydrogen bonding.
Liquids and Intermolecular Forces: Part 2 of 10 [8:58] by Dr. Mike Christiansen
In this video Dr. Christiansen will teach you about ion-dipole and dispersion forces (also called London or Van der Waals) forces.
Intermolecular Forces [6:58] by Fus Chemistry Videos
Types of Intermolecular Forces [8:06] by Fus Chemistry Videos
Solids and Liquids [7:28] by Bozeman Science
In this video Paul Andersen compares and contrasts the properties of solids and liquids. Solids have a more organized structure which can either be amorphous or crystalline. In liquids the intermolecular forces are lower and so the molecules can show translation. Some of the properties that can be observed in liquids are viscosity, surface pressure and volumes of mixing.
London Dispersion Forces [5:01] by Bozeman Science
In this video Paul Andersen describes the positive force intermolecular forces found between all atoms and molecules. As electrons are distributed unevenly it creates instantaneous dipoles which hold molecules together. This force even holds uncharged atoms (like Noble gases) together. London dispersion forces increase as surface area, molecule sizing and pi bonding increases.
Dipole Forces [5:01] by Bozeman Science
In this video Paul Andersen describes the intermolecular forces associated with dipoles. A dipole is a molecule that has split charge. Dipole may form associations with other dipoles, induced dipoles or ions. An important type of dipole-dipole forces are hydrogen bonds.
Intermolecular Forces [7:00] by Bozeman Science
In this video Paul Andersen explains how intermolecular forces differ from intramolecular forces. He then explains how differences in these forces account for different properties in solid, liquids and gases. Some of these properties include the boiling point, melting point, surface tension, capillary action and miscibility. Intermolecular forces between gas molecules creates variation from ideal gas law.
Introductory Level Videos
Polar & Non-Polar Molecules: Crash Course Chemistry #23 [10:45] by CrashCourse
Molecules come in infinite varieties, so in order to help the complicated chemical world make a little more sense, we classify and categorize them. One of the most important of those classifications is whether a molecule is polar or non-polar, which describes a kind of symmetry - not just of the molecule, but of the charge. In this edition of Crash Course Chemistry, Hank comes out for Team Polar, and describes why these molecules are so interesting to him.
You'll learn that molecules need to have both charge asymmetry and geometric asymmetry to be polar, and that charge asymmetry is caused by a difference in electronegativities. You'll also learn how to notate a dipole moment (or charge separation) of a molecule, the physical mechanism behind like dissolves like, and why water is so dang good at fostering life on Earth.
Liquids: Crash Course Chemistry #26 [11:03] by CrashCourse
In this episode of Crash Course Chemistry, Hank gives you the low down on things like London Dispersion Forces, Hydrogen Bonds, Cohesion, Adhesion, Viscosity, Capillary Action, Surface Tension, and why liquids are just... WEIRD!
Intermolecular Forces and Boiling Points [10:53] by Professor Dave Explains
Why do different liquids boil at different temperatures? It has to do with how strongly the molecules interact with each other. Find out all the different ways, and how to use them to make predictions about matter!
Intermolecular Forces [9:15] by The Chemistry Solution
This chemistry tutorial is designed to help compare and contrast commonly encountered intermolecular forces and interactions. The intermolecular forces that are discussed are ion-dipole interactions, dipole-dipole interactions, hydrogen bonding interactions and London dispersion forces.
Additional Useful Resources
Intermolecular Properties - web-based tutorials for GenChem at The University of Texas
Forces of Attraction - web-based tutorials for GenChem at The University of Texas
Practice
Forces of Attraction - link to webpage with link to worksheet and answer key for GenChem at The University of Texas
The Solid and Liquid States - Quiz bank for GenChem at Ohio State
"Click" on the appropriate "Quiz" button to select quizzes for practice from the following topics:
- Boiling Points
- Hydrogen Bonding