Ch 1. Essential Ideas
StudyWork = Study + Homework
In order to perform well on any quiz or test, I must:
In order to perform well on any quiz or test, I must:
- Read and study the class notes - Foundations of Chemistry
- Complete all worksheets or other work (i.e. ChemActivities) assigned and discuss all answers with my Pod.
- Read and study Chapter 1: Essential Ideas from openstax Chemistry (online) or downloaded from the following link:
| ch_1_openstax_chemistry.pdf | |
| File Size: | 2457 kb |
| File Type: | |
- Read and study Ch. 1 from General Chemistry by Ebbing and Gammon - as needed for my success.
- Read and study Ch. 1 thru Ch. 5 from Calculations in Chemisty - as needed for my success.
- Review the appropriate learning resources from this website- as needed for my success.
Additional Study Resources
Notes on Chemical Fundamentals from the National Math & Science Initiative (NMSI):
Notes on Chemical Fundamentals from the National Math & Science Initiative (NMSI):
| 01_chemical_foundations.pdf | |
| File Size: | 1009 kb |
| File Type: | |
The most important topics you will learn:
Scientific Notation
Did you know that the mass of the earth is 6000000000000000000000000000 grams?
... or that the mass of the common cold virus is 0.00000000000000000000003 grams?
Writing or saying such large (ginormous) and small (infinitesimal) numbers is subject to many, many typos and mistakes ! What are nurses supposed to do if they must write numbers much greater than 1 or much less than 1, as shown in the examples above ?
Scientific notation is a way to express very big and very small numbers with exponents as a power of ten. It is also sometimes called exponential notation. This week you will learn how to write ordinary (standard form) numbers in scientific notation and how to convert numbers in scientific into standard form.
The Metric or SI System of Measurement
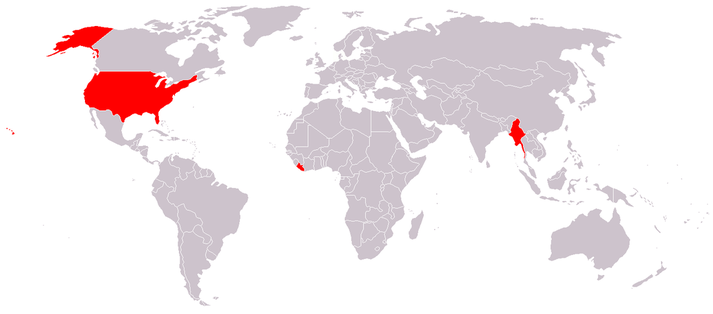
Did you know that according to the US Central Intelligence Agency's Factbook (2007), the International System of Units (SI, or metric system) has been adopted as the official system of weights and measures by all nations in the world except for Burma, Liberia and - get this - the United States ! *** Can you believe this? ***
Scientific Notation
Did you know that the mass of the earth is 6000000000000000000000000000 grams?
... or that the mass of the common cold virus is 0.00000000000000000000003 grams?
Writing or saying such large (ginormous) and small (infinitesimal) numbers is subject to many, many typos and mistakes ! What are nurses supposed to do if they must write numbers much greater than 1 or much less than 1, as shown in the examples above ?
Scientific notation is a way to express very big and very small numbers with exponents as a power of ten. It is also sometimes called exponential notation. This week you will learn how to write ordinary (standard form) numbers in scientific notation and how to convert numbers in scientific into standard form.
The Metric or SI System of Measurement
Did you know that according to the US Central Intelligence Agency's Factbook (2007), the International System of Units (SI, or metric system) has been adopted as the official system of weights and measures by all nations in the world except for Burma, Liberia and - get this - the United States ! *** Can you believe this? ***
This week you will learn about the advantages of the International System of Units (abbreviated SI from French: Le Système international d'unités) for manufacturing, trade, commerce, and, of course, medicine.
Significant Figures (SigFigs)
Let's say that you go to the clinic and have your weight measured by two different nurses - which of the following measurements makes most sense?
Nurse #1: Your weight = 135.682394 lbs.
... or
Nurse #2: Your weight = 135.7 lbs.
Remember This: Just because you can record a number does not indicate that the number has real meaning!
According to Nurse #1, your weight was recorded to the millionth of a pound - that's right to within 1/1000000 lb !! That means that Nurse #1 claims to be able to measure your weight to within a 0.000016 oz.
I mean REALLY?
The average adult has a lung capacity of about 6 liters so this means that one lung full of air weighs about 0.276 oz. Nurse #1's results only makes since if you were weighed while you were DEAD!! After all, Nurse #1 claims to be able to measure your breathing to less than 60 "milli-breaths" or to less than the weight of one hair!!
So you have to ask yourself: Are all of those numbers after the decimal useful ... or even real? Isn't the recorded weight by Nurse#2 much me reasonable and realistic? Can't you imagine that your weight might vary by a tenth (1/10) of a pound in a given day - as indicated by the "precision" of the measurement recorded by Nurse #2?
The concept of significant figures allows you to report ONLY those digits of a measured quantity that are reasonable and realistic.
Let's say that you go to the clinic and have your weight measured by two different nurses - which of the following measurements makes most sense?
Nurse #1: Your weight = 135.682394 lbs.
... or
Nurse #2: Your weight = 135.7 lbs.
Remember This: Just because you can record a number does not indicate that the number has real meaning!
According to Nurse #1, your weight was recorded to the millionth of a pound - that's right to within 1/1000000 lb !! That means that Nurse #1 claims to be able to measure your weight to within a 0.000016 oz.
I mean REALLY?
The average adult has a lung capacity of about 6 liters so this means that one lung full of air weighs about 0.276 oz. Nurse #1's results only makes since if you were weighed while you were DEAD!! After all, Nurse #1 claims to be able to measure your breathing to less than 60 "milli-breaths" or to less than the weight of one hair!!
So you have to ask yourself: Are all of those numbers after the decimal useful ... or even real? Isn't the recorded weight by Nurse#2 much me reasonable and realistic? Can't you imagine that your weight might vary by a tenth (1/10) of a pound in a given day - as indicated by the "precision" of the measurement recorded by Nurse #2?
The concept of significant figures allows you to report ONLY those digits of a measured quantity that are reasonable and realistic.
Conversions and Conversion Factors

Most of us know that the distance measurement of one (1) foot can be divided into 12 inches. And that one (1) meter can be said to be made up of 100 centimeters. But what about converting from a distance of kilometers (km) to millimeters - or even to inches or miles? How do we accomplish these types of calculations? The use of conversion factors will enable you to convert from any quantity and unit to any other quantity and unit. This process is known as dimensional analysis or unit-factor conversion.